
Finasteride, brand name Propecia™, is an FDA-approved oral medication for the treatment of hair thinning and hair loss in men caused by genetic factors referred to as androgenetic alopecia. The drug received FDA approval in 1997 and was obtainable by prescription by 1998. Under the brand name Propecia™ it is manufactured by Merck & Co. as 1mg tablet taken daily. Unlike minoxdil (Rogaine, Regaine), finasteride is only prescribed to men. Finasteride is also available under the brand Proscar™, but this is a 5mg tablet prescribed to reduce enlarged prostates.
How Finasteride (Propecia™) Works
Finasteride reduces the effect male hormone activity on susceptible hair follicles. In the case of male pattern hair loss, it inhibits an enzyme called 5-alpha reductase that triggers the conversion of the male hormone testosterone to dihydrotestosterone (DHT).
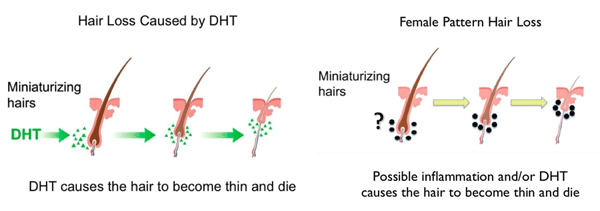
DHT is the primary cause of miniaturization of hairs for a group of men, otherwise known as hair thinning. Dihydrotestosterone disrupts the normal function of hair follicles that are susceptible to disruption by DHT. Not all hair is susceptible to DHT, but hair located at the hairline, top of the scalp, crown and the back of the head are affected by those suffering from pattern hair loss. Hair thinning or miniaturization is the process where hair naturally sheds (telogen phase of hair growth), then grows back (anagen phase), but each time it grows back it gets thinner in diameter. With each growth phase, hair comes back thinner until it eventually disappears. The thinnest hairs are known as vellus hairs, which are so fine that they are barely visible, and they don’t grow long (think of peach fuzz). Vellus hairs eventually disappear, which is true hair loss.
How Does DHT Cause Hair Loss?
A very important part of hair growth is located at the base of the hair shaft called the dermal papilla. Dermal papillae cells divide and differentiate to form new hair follicles. The dermal papillae are connected directly to blood vessels that carry the essential nutrients needed for healthy hair follicles and proper hair growth. The dermal papillae have many androgen (hormone) receptors which when exposed to DHT results in hair follicles’ growth process being disrupted.
During hair thinning, the hair growth cycle (anagen phase) shortens, and the resting stage (telogen phase) gets longer. Decrease in nutrient and metabolic process result in hair getting thinner, lighter and smaller. As hair becomes less healthy, they become light-colored, fine, and gradually become less noticeable. This is called vellus hair, which is commonly known as peach fuzz. Vellus hair is the last stage of hair miniaturization before it’s completely lost (hair loss).
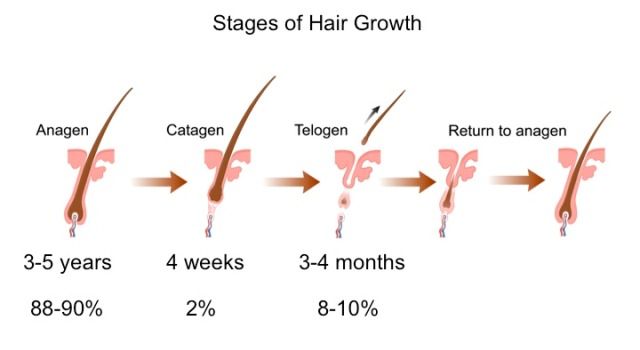
Normal hair growth has hair periodically shedding, then growing again. Hairs affected by DHT grow back thinner in every succeeding growth cycle.
Dihydrotestosterone (DHT) causes hair to growth back progressively thinner in a process called miniaturization.
Finasteride Stops DHT Conversion that Causes Hair Thinning in Susceptible People
Finasteride acts by reducing the amount of testosterone converted to DHT. By preventing DHT from forming within the scalp, finasteride prevents the DHT from initiating the process which results in androgenetic alopecia.
While all males and females naturally produce testosterone which converts to dihydrotestosterone (DHT) , not all possess hair that is affected negatively by DHT. DHT affects only certain hairs in certain people, which is why some men experience hair loss and some don’t, even though all men and women do produce DHT from testosterone. Those that don’t experience hair loss at by a certain age may experience it later years.
Some Hair is Genetically Resistant to DHT
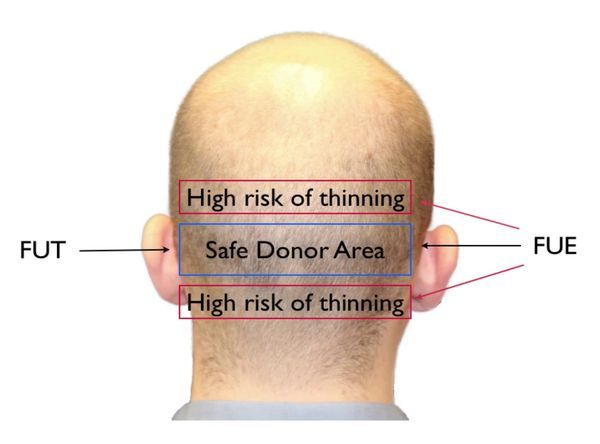
The safe or permanent zone is genetically resistant to DHT. Hair is taken from this area in follicular unit transplantation (FUT) hair transplants, but follicular unit extraction (FUE) transplants do take donor hair from outside this safe zone.
In most men including those who suffer from male pattern baldness, DHT does not affect hairs below in a small area below the crown, near the neck, and the lower sides of the head, otherwise known as permanent hairs or the permanent zone. This is why this area or genetically-resistant hairs to the effects of DHT is also called the donor area, which is used to take hair follicle grafts used in hair transplantation. In rare cases, even the permanent zone can be affected by male pattern baldness. But for almost all men, even with the most severe hair loss, this rim of hair (often called the horseshoe pattern) remains when the scalp and crown hair are lost.
Sexual Side Effects of Finasteride
There is a an ongoing fear of using finasteride to treat hair loss because of reports of sexual side effects, both immediate and long-term, associated with finasteride. The direct cause of finasteride to adverse sexual side effects has not been established, but the FDA did revise the warning label of finasteride in 2012 to include that sexual side effects were more common than first acknowledged when it was approved by the FDA for hair loss in 1997. When finasteride was first approved for hair loss in 1992, only 3.8% of patients in the trial study reported sexual side effects, but the FDA’s own study from 1998 to 2011 revealed this number to be higher at 14% from their sampling of 421 individual cases.
Another factor that caused the FDA to revise warning label of finasteride was reports of sexual side effects of finasteride continuing after use of the drug stopped. When the small number of patients first reported side effects prior to its 1997 FDA approval, sexual side effects were limited to early stages of the drug’s use, which would be discontinue after the drug was no longer taken. Subsequent reports after finasteride’s initial FDA approval found that side effects persisted for at least 3 months after use was discontinued, and in some reports, were permanent.
Sexual side effects associated with finasteride include:
- Decrease of sexual interest
- Decrease of sexual performance
- Erectile dysfunction
- Affected quality of semen
- Affected quantity of semen
- Infertility
Some Patients Don’t Respond to Finasteride
While many men refuse to take finasteride altogether because of the risk of sexual side effects, some young men (age group at highest risk of side effects) do take it out of desperation. However, some young men have come to Dr. Prasad’s practice with finasteride having virtually no effect on their progressive hair loss. While the science of blocking DHT conversion seems sound in theory, in practice it just doesn’t work on everyone. Initial studies of finasteride showed that 65% of men treated showed increased hair growth after 1 year, 80% of men after 2 years, and 77% of men after 5 years using actual difference of hair count. However, a separate, independent panel viewing photographs only showed visible results were seen in 48% of the same patients at 1 year, 66% after 2 years, and 48% after 5 years.
Hair Regeneration appears to Work “Better” than Finasteride, on More People, with No Sexual Side Effects
Natural-looking hair density without incidence of continued hair loss or thinning with the Hair Regeneration treatment clearly shows it works more effectively than finasteride for men who’ve been treated. Both patients shown here were taking finasteride regularly when their “before” images were taken. Images show significant hair growth, thickened hair, and a dense, natural-looking volume of hair came after the Hair Regeneration treatment with no surgical hair transplant performed. A synergy may be working where finasteride reduces DHT and Hair Regeneration stimulates hair growth.
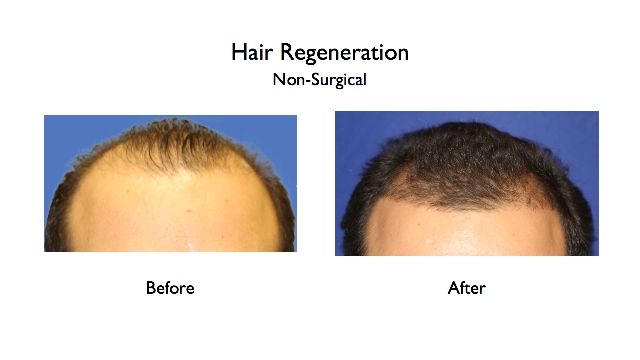
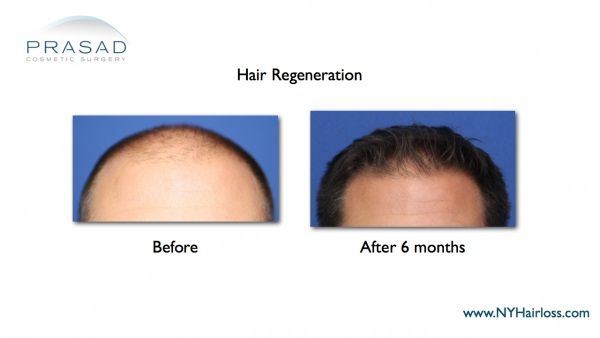
Two men in their 30s were on Propecia™ (finasteride) before receiving Hair Regeneration non-surgical treatment. Hair Regeneration results are dramatic, with no hair transplant surgery performed and no side effects experienced.
Hair Regeneration has visible results in 99% of male and female hair loss patients. Women are not generally prescribed finasteride although limited data appears to show some benefit in select groups of women.
