ACell + PRP Case Studies 2010-2011
Acell + PRP injected into the balding skin of a 70-year old man results in new hair growth.
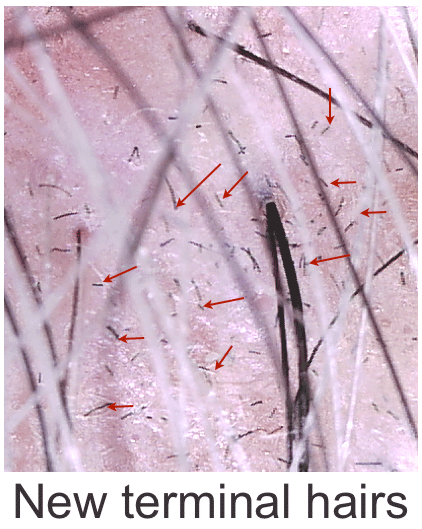
10x magnification shows new terminal hairs next to existing hairs after Hair Regeneration injection performed 3 months earlier.
Note two important details:
1) New terminal hairs sprouting all over the injected thinning area.
2) The new hairs are the original dark color — NONE are gray.
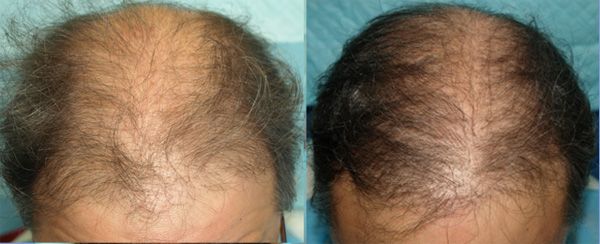
ACell + PRP Injection Results
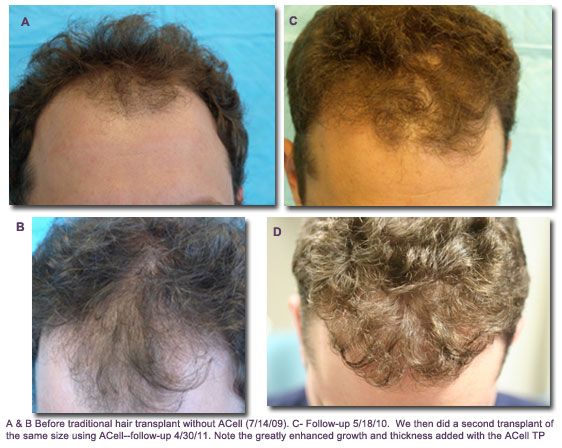
ACell Patient — Before & After Photos
Hair Regeneration Treatment
Follow up at 16 months. Patient will continue to be monitored. (No other future treatments scheduled currently)
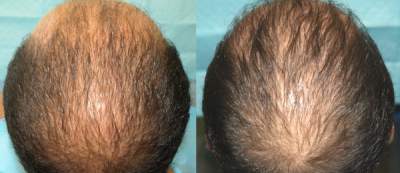
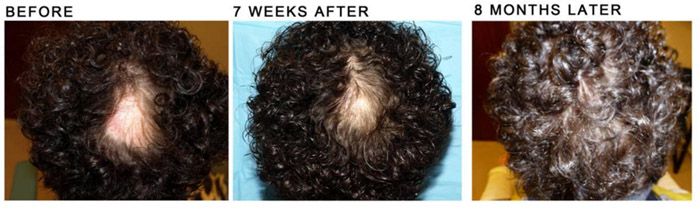
Patient aged 29-years old. Treatment: Acell + PRP
Condition: Progressive genetic hair loss. Patient did not want hair transplant surgery or oral treatment with Propecia.
Recommendation: Trial injections with Acell plus Platelet Rich Plasma (PRP)
Expectation: To possibly halt hair loss and evaluate in 6 months for future treatment or surgery.
Current evaluation: No further treatment until 1 year follow up.
Photos: (LEFT) Before 7/13/10 & After (RIGHT) 1/18/11
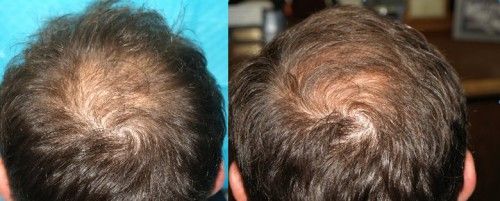
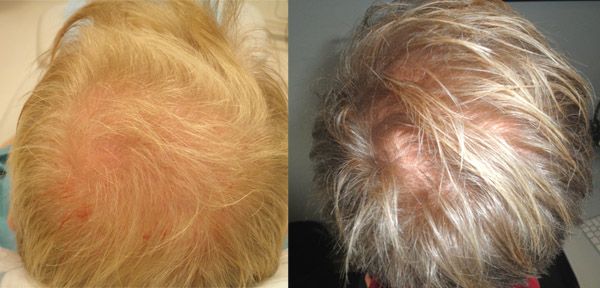
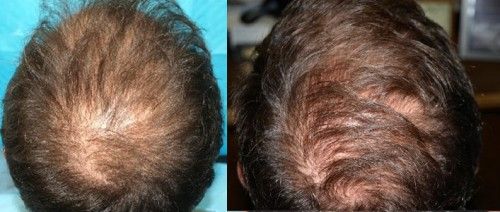
Patient aged 52-years old. Treatment: Acell + PRP
Condition: Class VI genetic hair loss pattern. Patient had previous hair transplant surgery with poor growth.
Recommendation: Trial injections with Acell plus Arterial Platelet Rich Plasma (APRP)
Expectation: To possibly halt additional hair loss and thicken existing hair transplants.
Current Evaluation: After a 1 year follow-up there was additional hair growth from the surrounding hair and from previous hair transplants. No apparent ongoing hair loss.
Photos: Before (LEFT) 1/16/10, After (RIGHT) 1/15/11
Hair Regeneration Treatment Depends on Customization for Individuals
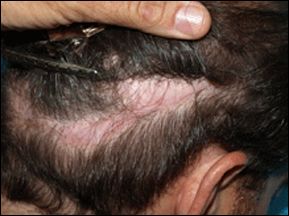
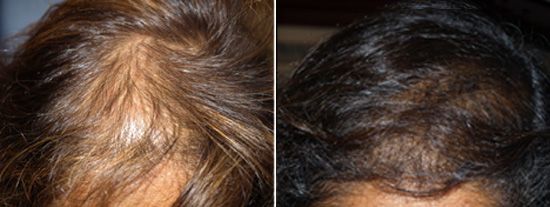
Scarring in old hair transplant donor areas before ACell treatment

New hair growth in donor scar 4 months later.
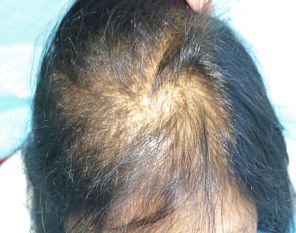
Female patient before
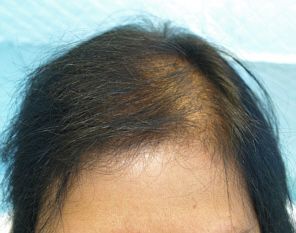
After 3 months post op grafts dipped in ACELL prior to transplanting
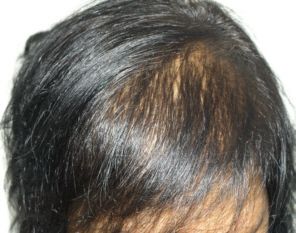
Female patient before
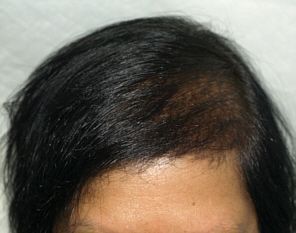
After 3 months post op grafts dipped in ACELL prior to transplanting
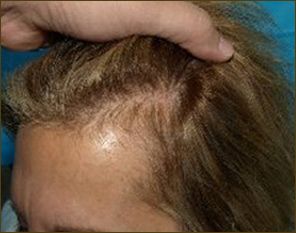
Female patient before
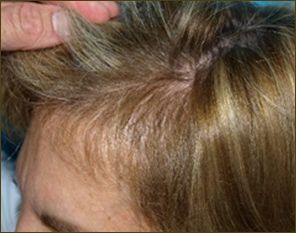
After 6 months post op grafts dipped in ACELL prior to transplanting
ACell’s technology is not limited to one market segment (e.g., wound care), but can serve as a core technology for treating a wide range of human medical conditions. For more information go to: www.acell.com
Current Results of Hair Regeneration, and Refinement of the ACell+PRP Algorithm
Work on ACell+PRP continually evolved after 2011. The Hair Regeneration treatment is not a specific formulation of ACell and PRP that applies to all patients, but rather an algorithm that determines the optimal formulation of ACell and PRP for each patient based on these variables:
- Gender
- Age of onset of pattern hair loss
- Degree of pattern hair loss
- Other medical factors, such as past hair transplant(s)
- Other medications used, such as finasteride
You can see more comprehensive case studies tracking patient results past 2011 here
If you have specific questions about the Hair Regeneration treatment, or want to know how it differs from other ACell+ PRP treatments, or would like to book a consultation, you can call us in Manhattan at (212) 265-8877, or at Garden City, Long Island at (516) 742-4636, or at Vienna, Virginia at (703) 356-1336, or fill out the contact form below, and we will get back to you.